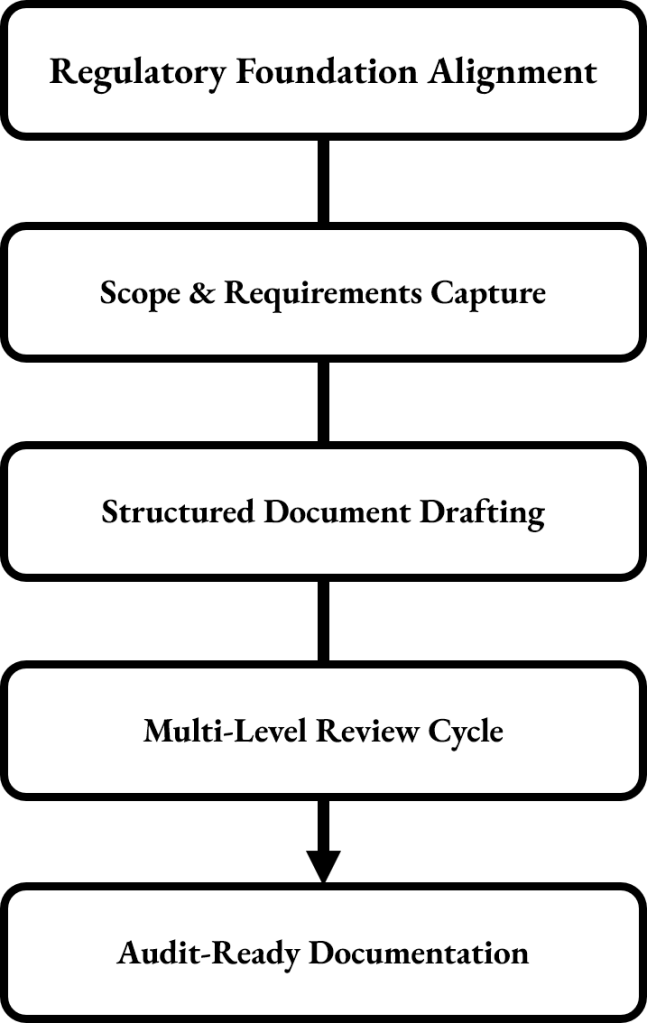
Stavigent provides audit-ready validation and Quality Assurance (QA) documentation. Using a structured drafting and review process based on GMP, Annex 11, and GAMP 5 principles, documents are created with clarity, consistency, and regulatory compliance, ensuring companies get high-quality deliverables that integrate seamlessly into existing quality systems.

Leave a comment