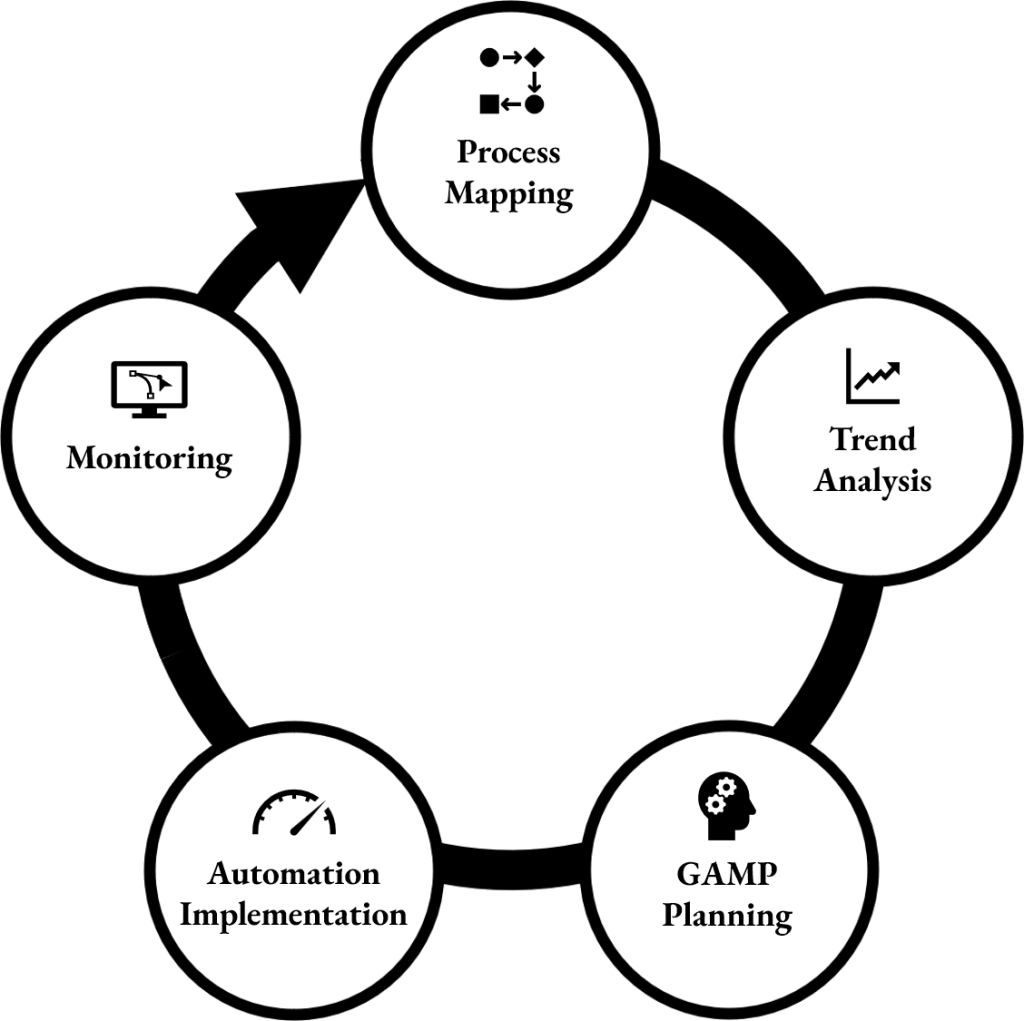
Continuous GMP & Digital Improvement
Map. Analyse. Plan. Automate. Monitor. Improve. Repeat.
Stavigent identifies manual, error-prone steps in GMP workflows and creates practical automation solutions to significantly reduce human errors.
GAMP-based planning ensures automation solutions follow risk-based validation principles defined in GAMP 5.
Automated & Streamlined GMP Processes
Effective automation begins with fully digital, well-defined processes, not with robotics or scripts.
Hybrid workflows involve manual handoffs, reconciliation processes, and data integrity risks that weaken automation efforts.
By transitioning to end-to-end digital workflows, organizations establish the foundation needed for reliable automation, advanced analytics, and robotics in GMP environments.
Source: Based on industry practices and regulatory guidance covered in Computerised System Validation: The GAMP 5 Approach (ECA Foundation & ECA Academy).

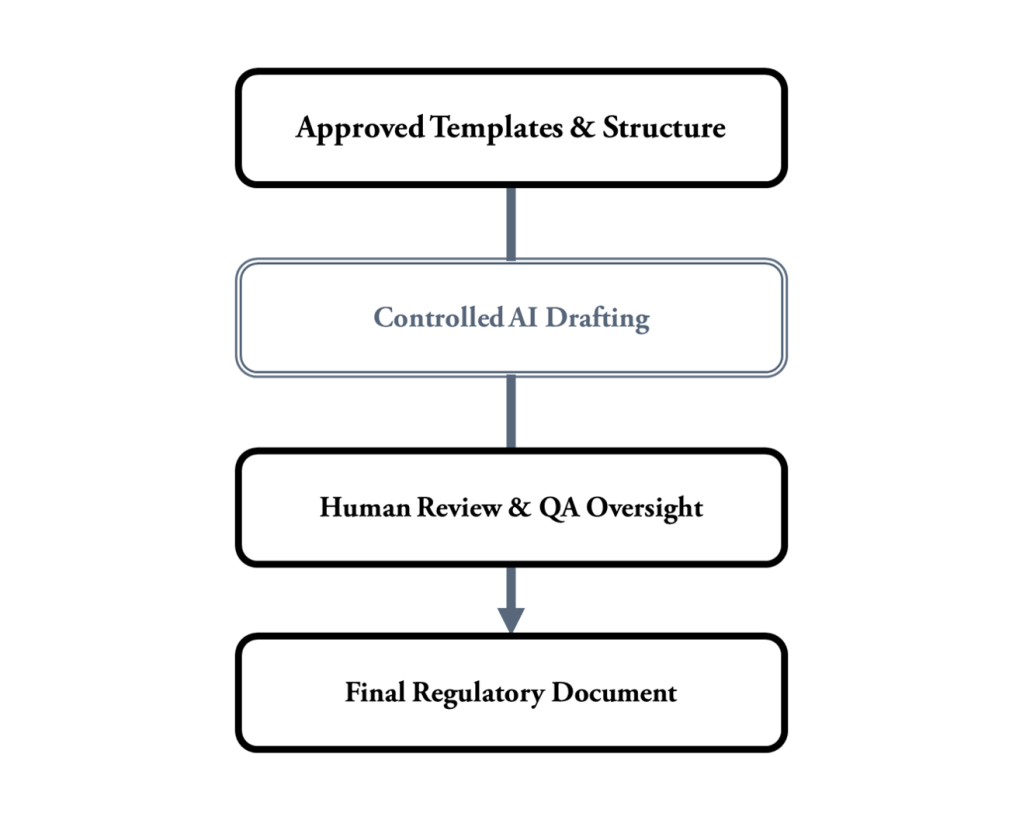
AI-Supported Regulatory Writing
Stavigent supports AI-assisted regulatory writing within controlled, GMP-compliant frameworks.
Instead of producing uncontrolled content, AI is integrated into structured templates, predefined prompts, and approved document frameworks to support consistent, compliant drafting.
The approach emphasizes speeding up initial drafts, standardizing language, and reducing repetitive manual work, all while maintaining human oversight and regulatory accountability.
By using standardized templates, controlled AI prompts, and review-ready structures, organizations can greatly reduce documentation efforts, enhance consistency across documents, and ensure full compliance with GMP, Annex 11, and GAMP 5 standards.
AI should serve as a drafting assistant, maintaining transparency, traceability, and audit readiness at all times.
Happy New Year from Stavigent! 🎉
May 2026 bring smoother processes and fewer deviations for all quality and validation teams.